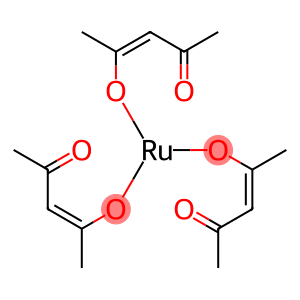
Ruthenium 2,4-pentanedionate
Ruthenium(III)-2,4-pentanedionate
CAS: 14284-93-6
Molecular Formula: C15H21O6Ru
Ruthenium 2,4-pentanedionate - Names and Identifiers
| Name | Ruthenium(III)-2,4-pentanedionate |
| Synonyms | Rutheniumpentanedionate Ruthenium 2,4-pentanedionate RUTHENIUM(III) ACETYLACETONATE Ruthenium(III) acetylacetonate Ruthenium(Ⅲ) 2,4-pentanedionate Ruthenium(III)2,4-pentanedionate Ruthenium iii 2,4-pentanedionate Ruthenium(III) 2,4-pentanedionate RUTHENIUM(III) 2,4-PENTANEDIONATE Ruthenium(III)-2,4-pentanedionate Tris-(2,4-pentanedionato)-ruthenium Tris(acetylacetonato)ruthenium(III) TRIS(ACETYLACETONATO)RUTHENIUM(III) Rutheniumacetylacetonateredbrownxtl Tris(acetylacetonato)ruthenium (III) Tris(2,4-pentanedionato)ruthenium(III) TRIS(PENTANE-2,4-DIONATO)RUTHENIUM(III) 2,4-PENTANEDIONE, RUTHENIUM(III) DERIVATIVE 4-pentanedionato-o,o')-tris((oc-6-11)-rutheniu ruthenium(3+) tris[(2Z)-4-oxopent-2-en-2-olate] Ruthenium(III) 2,4-pentanedionate~Tris(acetylacetonato)ruthenium(III)~Tris(2,4-pentanedionato)ruthenium(III) |
| CAS | 14284-93-6 |
| EINECS | 238-193-0 |
| InChI | InChI=1/3C5H8O2.Ru/c3*1-4(6)3-5(2)7;/h3*3,6H,1-2H3;/q;;;+3/p-3/b3*4-3- |
| InChIKey | RTZYCRSRNSTRGC-LNTINUHCSA-K |
Ruthenium 2,4-pentanedionate - Physico-chemical Properties
| Molecular Formula | C15H21O6Ru |
| Molar Mass | 398.39 |
| Melting Point | 260°C (dec.)(lit.) |
| Boling Point | 187.6°C at 760 mmHg |
| Flash Point | 71.9°C |
| Water Solubility | Soluble in most organic solvents such as acetone, chlorinated hydrocarbons, alcohols, cyclohexane and benzene. Insoluble in water. |
| Vapor Presure | 0.174mmHg at 25°C |
| Appearance | Red to purple powder |
| Color | White |
| Storage Condition | Store below +30°C. |
| MDL | MFCD00000030 |
Ruthenium 2,4-pentanedionate - Risk and Safety
| Risk Codes | 36/37/38 - Irritating to eyes, respiratory system and skin. |
| Safety Description | S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S36 - Wear suitable protective clothing. S24/25 - Avoid contact with skin and eyes. |
| WGK Germany | 3 |
| FLUKA BRAND F CODES | 10 |
| TSCA | Yes |
| HS Code | 28439000 |
Ruthenium 2,4-pentanedionate - Reference Information
| EPA chemical information | Information provided by: ofmpub.epa.gov (external link) |
| use | can be used to make heterogeneous catalysts. MOCVD precursor compound, homogeneous catalyst. For example, specific tritiation in the region where sodium borohydride and aromatic carboxylic acids are hydrolyzed. It can also be used as an enantioselective hydrogenation catalyst for acids such as aryl acrylic acid and aryl propionic acid. Catalytic hydrogenation of dimethyl oxalate to ethylene glycol under mild conditions. |
Last Update:2024-04-10 22:29:15
Supplier List
Spot supply
Product Name: Ruthenium(III) acetylacetonate Visit Supplier Webpage Request for quotationCAS: 14284-93-6
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Ruthenium(III)-2,4-pentanedionate Request for quotation
CAS: 14284-93-6
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 14284-93-6
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: Ruthenium acetylacetonate Request for quotation
CAS: 14284-93-6
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 14284-93-6
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
Product Name: Ruthenium acetylacetonate Request for quotation
CAS: 14284-93-6
Tel: 0086-13605431940
Email: xuli678323@126.com
Mobile: +86-13605431940
QQ: 56860987
Wechat: 13605431940
WhatsApp: 13605431940
CAS: 14284-93-6
Tel: 0086-13605431940
Email: xuli678323@126.com
Mobile: +86-13605431940
QQ: 56860987
Wechat: 13605431940
WhatsApp: 13605431940
Spot supply
Product Name: Ruthenium Acetylacetonate Visit Supplier Webpage Request for quotationCAS: 14284-93-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: Ruthenium(III) acetylacetonate Visit Supplier Webpage Request for quotationCAS: 14284-93-6
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Ruthenium(III)-2,4-pentanedionate Request for quotation
CAS: 14284-93-6
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 14284-93-6
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: Ruthenium acetylacetonate Request for quotation
CAS: 14284-93-6
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 14284-93-6
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
Product Name: Ruthenium acetylacetonate Request for quotation
CAS: 14284-93-6
Tel: 0086-13605431940
Email: xuli678323@126.com
Mobile: +86-13605431940
QQ: 56860987
Wechat: 13605431940
WhatsApp: 13605431940
CAS: 14284-93-6
Tel: 0086-13605431940
Email: xuli678323@126.com
Mobile: +86-13605431940
QQ: 56860987
Wechat: 13605431940
WhatsApp: 13605431940
Spot supply
Product Name: Ruthenium Acetylacetonate Visit Supplier Webpage Request for quotationCAS: 14284-93-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History
Ruthenium 2,4-pentanedionate
5,5-Dimethylhexan-2-one
4-苯甲氧基苯氧基乙酸
4-Methyl-3-nitro-benzenesulfonyl chloride
乙基叠氮乙酸酯溶液
1,4-Methano-1H-indene, octahydro-4-methyl-8-methylene-7-(1-methylethyl)-, (1R,3aS,4R,7S,7aR)-
RARECHEM LK HD C008
5,7-dichloro-1H-imidazo[4,5-b]pyridine
5,5-Dimethylhexan-2-one
4-苯甲氧基苯氧基乙酸
4-Methyl-3-nitro-benzenesulfonyl chloride
乙基叠氮乙酸酯溶液
1,4-Methano-1H-indene, octahydro-4-methyl-8-methylene-7-(1-methylethyl)-, (1R,3aS,4R,7S,7aR)-
RARECHEM LK HD C008
5,7-dichloro-1H-imidazo[4,5-b]pyridine




